Research Interests
Epigenetic Basis of Diet-Associated Human Diseases
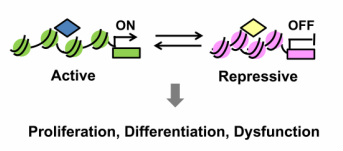
Epigenetics is the study of both heritable and
non-heritable changes in the regulation of gene activity and expression that
occur without an alteration in the DNA sequence, including DNA methylation,
histone modifications, and chromatin remodeling. Gene regulation can be achieved by alteration in
chromatin structure through controlling the conversion of inaccessible
heterochromatin to the accessible active euchromatin. This process, so called epigenesis, does not take place in only normal but pathological conditions. Abnormal transmission of epigenetic
information contributes to human pathology, such as cancer, aging,
developmental defects, metabolic disorders and various cardiovascular diseases. This dynamic
process has made an impact on the field of nutritional science as numerous studies
reported that changes in epigenetic state
may be induced by transient nutritional exposures or deficiency.
Our research has been interested in the understanding of the mechanistic basis by which epigenetic modifications regulate gene expression during environmental exposure and how alterations in epigenetic modifications link to diet-associated human diseases. Few biological questions we are interested in are:
1. How do parental nutrients stimuli pass on to the next generation by non-genetic means?
2. How do external nutritional stimuli connect to the histone modifiers activities or internal signaling pathways?
3. Whether dietary intervention could reverse the reprogramming caused by epigenetic changes?
Our research has been interested in the understanding of the mechanistic basis by which epigenetic modifications regulate gene expression during environmental exposure and how alterations in epigenetic modifications link to diet-associated human diseases. Few biological questions we are interested in are:
1. How do parental nutrients stimuli pass on to the next generation by non-genetic means?
2. How do external nutritional stimuli connect to the histone modifiers activities or internal signaling pathways?
3. Whether dietary intervention could reverse the reprogramming caused by epigenetic changes?
Current specific projects include:
1. The impact of the maternal nutrition on the offspring epigenome and the progression of cardiovascular disease in later life.
2. The role of Ikbkap-encoded IKAP protein in chromosome aneuploidy.
The mammalian ortholog of yeast Elp1, IKAP, is a component of the Elongator complex. The Elongator is a macromolecular complex (~900 kDa) comprising of two copies of a core complex, Elp1-3, and a sub-complex, Elp4-6. The complex was first identified by the copurification of the Elongator with RNA polymerase II (RNAP II) in budding yeast, and subsequently from human cells. Elp1 functions as a scaffold protein assembling the Elongator and is encoded by Ikbkap gene. Elp3, the catalytic subunit of Elongator, harbors a motif characteristic of histone acetyltransferases (HATs). Disruption of any subunits leads to loss of Elongator integrity. Indeed, deletion of Elp1 leads to down-regulation of Elp3, thereby affecting acetylation activity. Mutations to the human Ikbkap genes have been linked to the neurological disease familial dysautonomia (FD). Other than this, little is known about its biological functions, particularly in the context of whole animals. Recently, we generated germ cell-specific Ikbkap knockout mice, and demonstrated that Ikbkap is critically important for meiotic progression. Meiosis is the fundamental process that generates eggs and sperms required for sexual reproduction. In particular, female meiosis is highly error-prone in humans. Around 20% of all eggs contain abnormal chromosome numbers that are linked to spontaneous abortions, and developmental diseases like Downs Syndrome. Our lab interest is to determine the role of Elongator complex in oogenesis.
3. The Elongator in genomic integrity and tumorigenesis.
Since Ikbkap affects DNA repair pathway in male meiosis, and compromised DNA repair is a common feature of cancers, we are also determining the possible link of the Elongator to cancer.
4. Epigenetic factors in endothelial and lymphatic endothelial biology
5. Epigenetic factors in heart development
To achieve our research goals, we have developed a multidisciplinary approach that includes biochemical, cell biological and genetic analyses as well as the use of animal model.
1. The impact of the maternal nutrition on the offspring epigenome and the progression of cardiovascular disease in later life.
2. The role of Ikbkap-encoded IKAP protein in chromosome aneuploidy.
The mammalian ortholog of yeast Elp1, IKAP, is a component of the Elongator complex. The Elongator is a macromolecular complex (~900 kDa) comprising of two copies of a core complex, Elp1-3, and a sub-complex, Elp4-6. The complex was first identified by the copurification of the Elongator with RNA polymerase II (RNAP II) in budding yeast, and subsequently from human cells. Elp1 functions as a scaffold protein assembling the Elongator and is encoded by Ikbkap gene. Elp3, the catalytic subunit of Elongator, harbors a motif characteristic of histone acetyltransferases (HATs). Disruption of any subunits leads to loss of Elongator integrity. Indeed, deletion of Elp1 leads to down-regulation of Elp3, thereby affecting acetylation activity. Mutations to the human Ikbkap genes have been linked to the neurological disease familial dysautonomia (FD). Other than this, little is known about its biological functions, particularly in the context of whole animals. Recently, we generated germ cell-specific Ikbkap knockout mice, and demonstrated that Ikbkap is critically important for meiotic progression. Meiosis is the fundamental process that generates eggs and sperms required for sexual reproduction. In particular, female meiosis is highly error-prone in humans. Around 20% of all eggs contain abnormal chromosome numbers that are linked to spontaneous abortions, and developmental diseases like Downs Syndrome. Our lab interest is to determine the role of Elongator complex in oogenesis.
3. The Elongator in genomic integrity and tumorigenesis.
Since Ikbkap affects DNA repair pathway in male meiosis, and compromised DNA repair is a common feature of cancers, we are also determining the possible link of the Elongator to cancer.
4. Epigenetic factors in endothelial and lymphatic endothelial biology
5. Epigenetic factors in heart development
To achieve our research goals, we have developed a multidisciplinary approach that includes biochemical, cell biological and genetic analyses as well as the use of animal model.
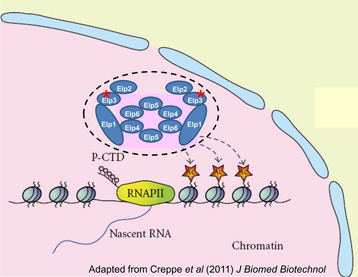
Functions of the Elongator.
In the nucleus, the Elongator mediates Histon H3 acetylation, and interacts with hyperphosphorylated RNAPII to contribute to transcriptional elongation.
All rights reserved. © 2015 FJ Lin